Email: cspc@cspc.cn
Email: cspc@cspc.cn
October 25, 2022
A study conducted by the team of CSPC and Prof. Luo Zhiguo from the Fudan University Shanghai Cancer Center, titled Pegylated liposomal doxorubicin combined with ifosfamide for treating advanced or metastatic soft tissue sarcoma: a prospective, single-arm phase II study, was successfully published in the journal of Clinical Cancer Research (IF: 13.801, JCR Division: Medical Division 1).

Background
Doxorubicin (ADM), in combination with Ifosfamide (IFO), is one of the standard treatment regimens for soft tissue sarcoma (STS). However, the toxicity of the combination therapy limits its application1. Studies in other tumor types have shown that pegylated liposomal doxorubicin (PLD) has comparable efficacy and better safety than ADM2. This study aimed to explore the efficacy and safety of PLD in combination with IFO in advanced or metastatic STS.
Method
The clinical study is single-arm, open-label and single-center that enrolled previously untreated patients with advanced STS and treated them with PLD (30 mg/m², d1) in combination with IFO (1.8 g/m², d1-5) every 21 days until the completion of 6 cycles of treatment, disease progression, development of intolerable toxicity, or subject death, whichever occurs first. The primary endpoint of the study was progression-free survival (PFS), with secondary endpoints covering overall survival (OS), objective response rate (ORR), safety, and biomarker exploration.
Results
From May 2015 to November 2019, 69 subjects were enrolled in this study, all included in the efficacy and safety analysis. The baseline characteristics are shown in Figure 1.
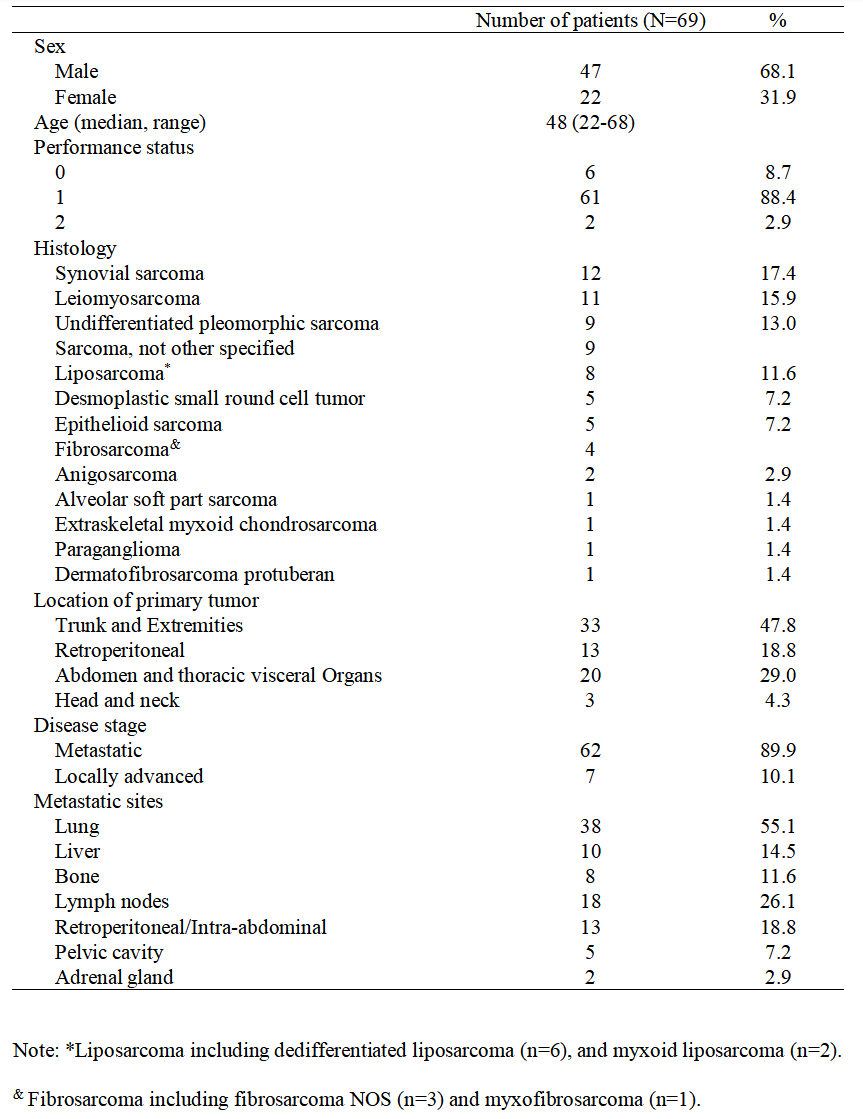
Figure 1: Baseline Characteristics
The median follow-up time was 47.2 months, with median PFS and OS of 7.3 (95% CI:5.7-8.9) and 20.6 (95% CI:16.3-25.0) months, respectively (Figure 2). One subject achieved a complete response, and 17 achieved a partial response, with ORR and disease control rate (DCR) of 26.1% and 81.2%, respectively.
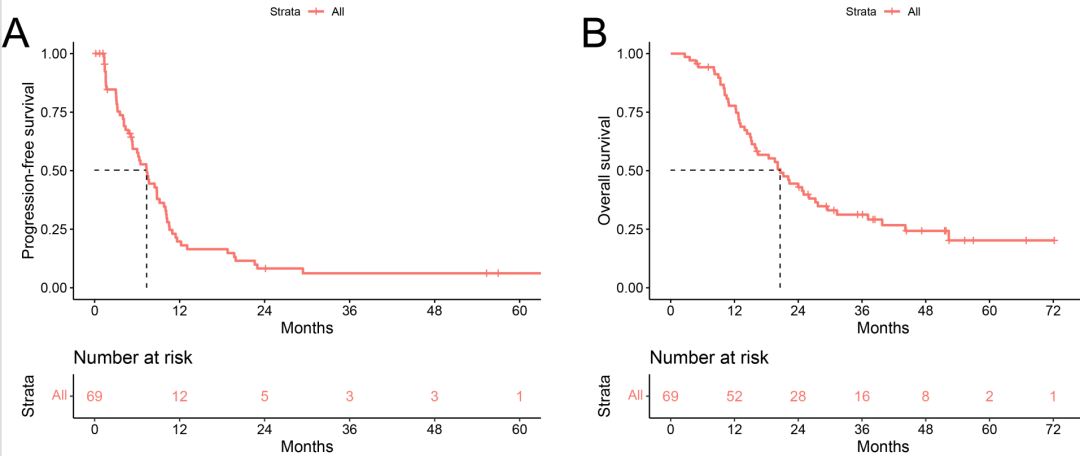
Figure 1: Baseline Characteristics
The toxicity of this regimen was controllable, with no Grade 3-4 cardiotoxicity observed. There was no significant change in left ventricular ejection fraction at baseline and after treatment (p=0.669). Grade 3/4 adverse events with an incidence of ≥10% included decreased neutrophil count (n=43, 62.3%), decreased white blood cell count (n=39, 56.5%), anemia (n=12, 17.4%), and febrile neutropenia (n=8, 11.6%).
Exploratory biomarker analysis showed that NF1 single nucleotide variants were associated with poorer OS (p<0.0001) and PFS (p=0.044). Negative Homologous Recombination Deficiency (HRD) was associated with improved OS compared to patients with positive HRD (p=0.0056).
Conclusions
This is the first Phase II study to investigate the use of PLD in combination with IFO for the first-line treatment of advanced STS. The results illustrate that PLD+IFO, showing similar efficacy and lower toxicity than the previously reported ADM-IFO, is an effective and well-tolerated treatment option for advanced STS.
References1. Guidelines Working Committee of the Chinese Society of Clinical Oncology (CSCO). Guidelines For the Diagnosis and Treatment of Soft Tissue Sarcoma 2022 [M]. People's Medical Publishing House (PMPH), 2022: 4.
2. O'Brien ME, Wigler N, Inbar M, et al. Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl(CAELYX/Doxil) versus conventional doxorubicin for first-line treatment of metastatic breast cancer [J]. Ann Oncol, 2004, 15 (3):440-449.